Those with concerns over or interests in genetically modified (GM) crops will still be reeling from the news released last week about a long-term study on rats to evaluate Monsanto’s herbicide-resistant GM maize (corn), along with its companion herbicide Roundup (glyphosate). The French and Italian researchers at the University of Caen discovered that typical levels of exposure to GM maize and Roundup caused cancer and premature death.
For those of us with long-standing concerns about the safety of GM crops, is this the study we’ve been waiting for – one that finally exposes GM crops for what they are? Or do we need to be cautious in how we interpret the results? And what about the flurry of criticism the study has created, especially from pro-GM quarters?
Why is the study important?
At the outset, it’s vital to note that this was the first-ever lifetime animal feeding study [1] of Monsanto’s GM maize and Roundup, the world’s most widely used herbicide. Prior studies – many carried out or funded by the products’ manufacturers – have only looked at the effects of 90 days’ ingestion. If we assume that humans live for an average 75 years and that rats live for 2 years, a 90-day rat feeding experiment is equivalent to less than 10 years of a human lifespan! Once approved for public sale, many people will eat GM food every day of their lives, and it’s a shocking comment on the corruption within the GM sector that lifetime animal feeding studies are not the norm prior to approval.

The paper that has sparked controversy about the safety of Monsanto’s GM maize and the world’s most widely used herbicide, Roundup
What does the study say?
The authors compared the effects of feeding rats with Monsanto’s hybrid, ‘Roundup Ready’ GM corn, named NK-603 – cultivated with and without Roundup – and normal, non-GM corn. They also studied how Roundup alone affected the rats, by adding it to their drinking water. In all cases, the authors were careful to use levels of the GMO corn and Roundup that were well below legal, supposedly ‘safe’ limits of exposure.
At the end of the 2-year study period, the authors reported some startling results:
- Up to 50% male rats and up to 70% female rats fed GM corn died during the treatment period, compared with 30% and 20%, respectively, of control rats fed a ‘substantially equivalent’ non-GM corn cultivar
- By the end of the experiment, the mortality rate of female rats given GM corn or Roundup (treated rats) was 2–3 times greater than that of controls – and they died earlier
- Female rats exposed to Roundup via drinking water demonstrated signs of endocrine system disruption and died earlier than control females. A similar effect was not observed in males
- Mammary tumours were the most frequently observed pathology in treated female rats
- Treated male rats displayed rates of liver abnormality that exceeded those of control males by up to 5.5 times
- No dose–response relationship was noted between the level of GM material in the diet and pathological symptoms, suggesting a ‘threshold effect’ – i.e. that presence of GM above a certain level triggers biological changes
Are there any problems with the study?
If the study’s methodology can be regarded as sound, these results are deeply disturbing. Monsanto’s GM maize is widely consumed by both humans and animals, and its companion herbicide, Roundup, is the most commonly used herbicide in the world. But who’s in a position to judge? As sure as night follows day, the pro-GM brigade has been out in force since the results were published, loudly criticising the study for a range of methodological flaws.
However, some of the criticism has come from sources with no obvious connection to industry.
After examining the paper closely ourselves, we have our own reservations about certain aspects:
- Rat strain used in study. Were the Sprague-Dawley (S-D) rats used in the study the most appropriate to select in a lifetime/2-year trial, given the extreme tendency for these rats to develop tumours in later life [2–4]? While the study aimed to show differences between control and treatment groups, it remains uncertain just how statistically and biologically relevant these differences were (see below). The authors demonstrated an apparently consistent trend for treatment groups to suffer rates of cancer and premature death that were greater than controls, but this must be seen in the context of a rodent strain that is already highly predisposed to cancer, especially in later life. Scientifically, if not ethically, the study is screaming out to be repeated using the same, and one or more different, rodent strains. That said, at 14 months – around half-way through the study and the approximate mid-point of the rats’ lifespan – no untreated animals had any signs of tumour, whereas 10–30% of treated females in all but one group had developed tumours. This is powerful evidence that both Roundup Ready corn and Roundup itself are cancer-promoting in rats, and possibly in humans
- Sample size of 10. Coupling the genetic tendency of Sprague-Dawley rats to develop cancer in later life with the small sample sizes (10 per treatment or control group), you have a recipe for apparent results being generated by chance alone. While the small sample size has been criticised for preventing firm conclusions from being drawn, others have pointed out that 10 rats per group is probably OK for this purpose. But that doesn’t excuse not doing statistics! The sample size used in the current study is actually the same as that used in the regulatory assessment of NK-603 corn – as performed by...Monsanto!
- Presentation of results and statistical analyses. Strangely enough, if the researchers were looking for a slam dunk showing just how important their results were, they certainly haven’t presented them in a particularly logical way, nor have they analysed them adequately. Most importantly, they completely omitted to provide any statistical comparisons in some critical areas, such as the pathological findings in different organs from treated rats and controls (Table 2). We see this as a weakness not only in the authors original manuscript, but also of the editorial and peer-review quality of the journal, Food and Chemical Toxicology
- Rat diets. In evaluating the paper, there isn’t enough information available to guarantee that the rat chow provided to the control rats was completely GM free, given that the majority of the world’s animal feed now contains varying proportions of GM maize or soya. The study involved adding the GM maize, Roundup or GM maize plus Roundup to the standard rat chow, A04 (Safe, France). This inability to secure guaranteed GM-free rodent chow has been a long-standing problem with feeding studies of GM crops, and attempts to change the control rat diet from the standard rat chow and then make comparisons with other studies complicate the interpretations
- Possible confounding effects of bisphenol A (BPA). It appears that the rats were kept in polycarbonate cages, and there is no indication whether water was delivered in plastic or glass bottles. However, it is known that BPA can leach both from polycarbonate cages and from plastic water bottles, which may influence the results, particularly because of BPA’s well-known endocrine-disrupting effects
- Declaration of interests. The authors declare that they have no conflicts of interest at the end of the paper. But, while there may not have been any direct financial conflict of interest at the time of publication, we suggest that it would have been more transparent for the lead author, Professor Gilles-Eric Séralini, to declare his role as President of the Scientific Council of CRIIGEN. CRIIGEN is, “An independent non-profit organization of scientific counter-expertise to study GMOs, pesticides and impacts of pollutants on health and environment, and to develop non polluting alternatives.” It is also relevant that Prof Séralini has written a book about the study and its implications, entitled Tous Cobayes! (All Guinea Pigs!), that is being published today by Flammarion. A film by Jean-Paul Jaud, adapted from the book, will also be launched
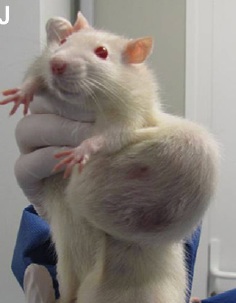
- Photographs. Comprehensive and gruesome pictures of the tumours occurring in treated rats are provided in Figure 3, but comparison pictures from untreated rats are entirely absent. Ultimately, it is the comparison between treatment and controls that allows conclusions to be drawn

Photographs of Sprague-Dawley rats in the study: J = GM maize alone; K = GM maize + Roundup; L = Roundup alone [1]. But how old were these rats and what about control group rats?
The fall-out continues
While unlikely in itself to ever be regarded as conclusive evidence of the carcinogenicity of Monsanto’s GM maize and Roundup products, this study is likely to trigger a sea change in approaches to the evaluation of GM.
Discussions about the relevance of the findings have already been held in the European Parliament, led by French lawyer, previous CRIIGEN president and now Member of the European Parliament (MEP), Corinne Lepage.
The controversial European Food Safety Authority (EFSA), well known for its track record of green-lighting European GM crop registrations, has already been asked by the European Union executive to evaluate Séralini’s study. EFSA’s response is likely before the end of the year, following a preliminary review that is due to report in early October. There’s also been discussion about a repeat or counter study. But Prof Séralini has been quick to say that, “It's out of the question that those who authorised (Monsanto's) NK603 carry out a counter-study of our findings as there’d be a conflict of interest”.
Conclusions
Although there are, without doubt, several questionable aspects to this study [1], which we hope the authors will address in future publications, the results remain of profound concern. Most importantly, the study attempted to investigate the effect of GM maize and Roundup across the entire lifespan of the rat, at low levels of exposure that include the dose range to which humans are typically exposed. Any signal of potential carcinogenicity at these relatively low levels of exposure, as identified in any rodent model, must be taken seriously – and this study appears to have uncovered plenty such signals. Among these are the endocrine disruptive effects of Monsanto’s Roundup, the herbicide used in conjunction with the Roundup-resistant GM maize. Russia is certainly taking the results seriously.
We wholeheartedly agree that the researchers should continue to refuse to let EFSA examine their findings, as we share their concern that EFSA would simply dismiss the results.
More long-term feeding studies are definitely needed, and different strains of rodent must be used. The Séralini study [1], among other things, shows up the gross inadequacy of the 90-day studies presently carried out by the biotech industry to ensure regulators approve their patented crops.
We also believe it would be in everyone’s interest for the journal to issue a minor amendment, via an erratum, giving appropriate calculations of statistical significance. If we’re going to rely on science to make our decisions, let’s have good science. It’s one of the few tools we’ve got to help us make decisions that affect the future of humankind and the other animals and plants with which we share the planet.
Regardless of the results of the present study, we feel that an immediate worldwide moratorium on the cultivation of GM crops is essential, based on current evidence of their potential health dangers. And it remains an insult to our fellow human beings that GM-containing products are not universally labelled.
Truly, the Californian vote on GM labelling is becoming the issue of our times.
Please circulate this article widely.
References
[1] Seralini G-E et al. Food Chem Toxicol 2012 Sep 11. pii: S0278-6915(12)00563-7. doi: 10.1016/j.fct.2012.08.005. [Epub ahead of print]
[2] Davis RK et al. Cancer Res 1956;16:194–7.
[3] Prejean JD et al. Cancer Res 1973;33:2768–73.
[4] Durbin PW et al. Cancer Res 1966;26; 400–11.







Comments
your voice counts
26 September 2012 at 11:50 pm
However 'flawed' the study may have been statistically in the eyes of the purists, so as to be fully acceptable to them, the very fact that we see these tumours, that they occurred at earlier times, that the rats died earlier etc. is quite enough for the non-scientific consumer to change habits.
Unfortunately, the consumers may never find this out. My comment in the UK Telegraph mentioning this on a topic regarding deformed babies, was erased in minutes by the censors!!
Soy, even non GM soy, contains high levels of phytic acid in soy reduce assimilation of calcium, magnesium, copper, iron and zinc. Phytic acid in soy is not neutralized by ordinary preparation methods such as soaking, sprouting and long, slow cooking. High phytate diets have caused growth problems in children.
• Trypsin inhibitors in soy interfere with protein digestion and may cause pancreatic disorders. In test animals soy containing trypsin inhibitors caused stunted growth.
• Soy phytoestrogens disrupt endocrine function and have the potential to cause infertility and to promote breast cancer in adult women.
• Soy phytoestrogens are potent antithyroid agents that cause hypothyroidism and may cause thyroid cancer. In infants, consumption of soy formula has been linked to autoimmune thyroid disease.
• Vitamin B12 analogs in soy are not absorbed and actually increase the body's requirement for B12.
• Soy foods increase the body's requirement for vitamin D.
• Fragile proteins are denatured during high temperature processing to make soy protein isolate and textured vegetable protein.
• Processing of soy protein results in the formation of toxic lysinoalanine and highly carcinogenic nitrosamines.
• Free glutamic acid or MSG, a potent neurotoxin, is formed during soy food processing and additional amounts are added to many soy foods.
• Soy foods contain high levels of aluminum which is toxic to the nervous system and the kidneys.
Corn? Even non GM corn contains a yet-unidentified allergen, rhat inflames 3 specidfic body parts, (abdominal mass, B cells. vein valves). An inflamed body part allows easy entry for the cancer nucleus and cancer complex to start and fuel a malignancies at that site, in the presence of other 'players'. Clark HR PhD ND 2007. Dr Clark's works since 1995 are based on over half a million repeatable, bio-resonance tests. She is not 'recognised' by mainstream medicine as no now-exposed-as-corrupt med journal would allow a natural, alternative, low profit cancer cure to be published.
Humans can choose. Poor animals can't.
Your voice counts
We welcome your comments and are very interested in your point of view, but we ask that you keep them relevant to the article, that they be civil and without commercial links. All comments are moderated prior to being published. We reserve the right to edit or not publish comments that we consider abusive or offensive.
There is extra content here from a third party provider. You will be unable to see this content unless you agree to allow Content Cookies. Cookie Preferences