On 16 May, the European Food Safety Authority (EFSA) closes its consultation on its draft opinion on vitamin D. The EFSA’s opinion will in effect set EU-wide dietary reference levels for the sunshine vitamin, these likely being applied equally by health authorities in sun-starved Scandinavia and sun-drenched Mediterranean countries. These levels are likely to be taken by national health authorities as the levels required where there is no vitamin D synthesis from sunlight exposure.
Worse than this, if these levels get set in stone, there is a real risk that health authorities will increase their determination to rid health store shelves of products containing 25 mcg (1000IU)/day or higher levels. In view of public health and the known science, this would be completely unacceptable.
There are numerous problems with the draft EFSA opinion. Most of them duplicate the many deficiencies linked to last year’s UK advice from the Scientific Advisory Committee on Nutrition (SACN) which we highlighted in our consultation response at the time.
The bottom line is that while it’s good to see health authorities like EFSA and the SACN now working out their recommended intakes based on circulating levels in the blood (serum), the levels that are considered adequate are far too low. In the case of EFSA, the target circulating level of 25-hydroxyvitamin D (also referred to as 25(OH)D or calcidiol) is 50 nmol/L (or, in the units more commonly used in the USA, 20 ng/ml).
This level is a massive two and a half times lower than the serum level recommended by vitamin D experts who have come together under the banner of the Vitamin D Council. EFSA, like so many health authorities before has developed its opinion based only on levels that prevent vitamin D deficiency diseases such as rickets and osteomalacia, the former applying children, the latter to adults, these being conditions resulting from inadequate bone mineralisation. EFSA excludes consideration of extensive research on vitamin D’s capacity to reduce the risk of a wide range of other diseases.
A very comprehensive study published earlier this month in the peer reviewed journal PLoS One shows clearly that doubling the circulating levels of vitamin D from the EFSA’s proposed level of 50 nmol/L (= 20 ng/ml) to 100 nmol/L (= 40 ng/ml) results in a startling greater than 65% reduction of “all invasive cancers combined”.
Another study, published in November 2015, showed that those with higher levels of circulating levels of 25-hydroxyvitamin D had a 21% reduced risk of developing kidney cancer.
The catastrophic issue here is that EFSA believes there are insufficient data to consider any studies or clinical data linked to deficiency diseases other than rickets and osteomalacia. EFSA typically works in a vacuum reviewing only what it considers to be “generally accepted data”. This same principle is also used by the FDA in the USA, where it is referred to as “significant scientific agreement”. EFSA ignores a huge amount of recent research as well as decades of clinical experience with doctors and other practitioners who routinely use vitamin D to support the health of patients. It ignores the increased requirement for vitamin D among the growing number of people who are overweight or obese, or those that use sunscreens and so impair their ability to synthesise vitamin D following sunlight exposure to the skin.
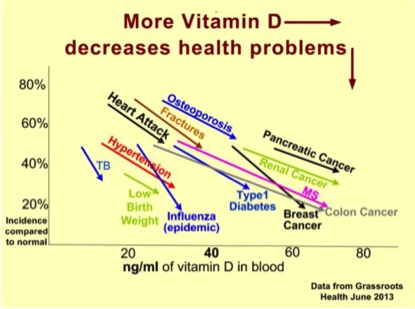
The fact is that oral vitamin D as a food supplement at daily dosages of at least 50 mcg (2000 IU) and preferably double this amount is one of the most cost-effective solutions to reducing the risk of a gamut of health problems or diseases. These not only include various cancers (e.g. breast, pancreas, kidney), but also heart attack, type 1 diabetes, hypertension, osteoporosis and bone fractures (see graphic below based on.
Source: Vitamin D Wiki (based on data from GrassrootsHealth, June 2013)
If EFSA doesn’t change the dietary reference intakes in its current draft report, there is a real risk that EU health authorities will start to declare levels significantly above the 15 mcg (= 600 IU) per day mark as medicinal. This will not only be very bad for all of us who have become reliant on high doses of oral vitamin D for our health – it will be especially bad for future generations who will be denied the higher doses many of us have benefited from.
What you can do
EFSA’s consultation is open to anyone and everyone in the EU. If you have benefited from dosages of vitamin D over 15 mcg (= 600 IU), it would be very helpful for you to provide a consultation response to EFSA before 16 May. Responses from healthcare practitioners, practitioner associations and nutrition educators would be particularly valuable.
Responses should be made online, following the guidelines given by EFSA.
We would suggest your response include at least:
- A reminder to EFSA that oral Vitamin D is a proven supplement that is highly cost effective and reduces the risk of a wide range of conditions including various cancers, heart attack, hypertension, diabetes, osteoporosis and bone fractures
- Indicate the daily amount you have taken or currently take (e.g. 100 mcg = 4000 IU/day, which is nearly seven times greater than the amount proposed for the adult dietary reference intake for adults)
- Specify if you have had blood tests for vitamin D and what levels you have attained
- Propose that the target levels for circulating 25-hydroxyvitamin D for adults should be set at 125 mmol/L (50 ng/ml), in line with the recommendations of the Vitamin D Council and an extensive Polish study
- Suggest that EFSA should follow the reference ranges for circulating 25-hydroxyvitamin D given by the Vitamin D Council, which are as follows:
- Deficient: 0-100 nmol/l (0-40 ng/ml)
- Sufficient: 100-200 nmol/l (40-80 ng/ml)
- High Normal: 200-250 nmol/l (80-100 ng/ml)
- Undesirable: > 250 nmol/l (> 100 ng/ml) Toxic: > 375 nmol/l (> 150 ng/ml)
Please remember the deadline for submission of your consultation response is 16 May!








Comments
your voice counts
21 April 2016 at 4:22 am
I take 4,000 IU of vitamin D per day in summer and 6,000 IU/day in winter in order to achieve a 50ng/ml blood serum level. You are recommending a limit that is much, much lower than I need to achieve a blood serum level that will be beneficial to my health in many ways, according to the Vitamin C Council. Please familiarize yourself with their work and significantly increase the recommended Vitamin D levels.
21 April 2016 at 10:18 am
What form of Vitamen D are you referring to?? Vit D2 a synthetic form of the vitamin often replaces real Vit D3 , but its effectiveness is poor. Not only are most people, especially dark skinned people, deficient in Vit D3 due to the lack of sunshine and the reduced exposure to it. There is no consideration given to the therapeutic value of Vit D3 . The use of statins is another cause of a reduction in Vit D3 due to the effect of reducing cholesterol production which impacts negatively on the mitochondria as well as digestion, steroid hormone production and bone formation. Nagalase is an enzyme produced by viruses as well as cancerous cells. Vit D3 is required to activate macrophages against nagalase if the patient is to have any chance of survival against a metastasizing cancer. High levels of Vit D3 in any chronic disease process is an absolute must. If these therapeutic levels are denied the numerous victims, it is obvious that the draconian low vitamin levels permitted amount to a deliberate form of malnutrition designed to not just destroy the patient but also to make the general population Vit D3 deficient with the obvious increase in chronic diseases. This is clearly not a patient serving agenda but a self-serving one which is also evident when one looks into the low levels of nutrients as recommended by the WHO's Codex Allimentarius.
21 April 2016 at 12:18 pm
I take 5,000ius daily and have been for the past two or three years and have only gained much better health because of it. I honestly have not had a cold or infection for all the time I started taking my Vitamin D supplement and have never felt better!
It should be people's right to benefit from Vitamin D supplementation, if they need it and many peoples' levels are way too low and the only way they can increase these levels to healthy amounts is to start taking high dose Vitamin D. I do agree though that people should have their levels tested first and then, thereafter, have them checked regularly (If they see the need) to make sure they are not getting 'too' much. I have heard of many people taking at least 10,000ius daily with no adverse health problems, so to reduce levels people can take so dramatically, is wrong and is only going to contribute to much poorer health of the general public, especially the ones who live in the Northern hemisphere and do not get much sunlight.
21 April 2016 at 5:18 pm
After over a year of complaining of severe fatigue and pain in my joints, not to mention the fact that my fingers started curving, a blood test finally revealed that I was extremely deficient in Vitamin D. I could not get an appointment to see my GP for three weeks so I ended up buying the Vitamin D3 supplements and within just a few days, I felt so much better. If this goes through, people won't have this option. Such is the influence of the pharmaceutical industry who are striving to ensure that any natural remedy is no longer easily available. Most concerning is that Vitamin D deficiency is common in countries like the UK and parts of Europe where we do not get much sunshine. Time to put people's health and wellbeing before the vested interests of corrupt individuals.
21 April 2016 at 7:22 pm
I and my family take vitamin D3 2200 IU - no colds or flu, plenty of energy.
21 April 2016 at 9:03 pm
Thank you for this article. Email sent to EFSA.
21 April 2016 at 10:55 pm
I want to decide what I choose to put in my body myself!
Vitamin D in large doses - helps for a lot of things.....
22 April 2016 at 1:21 am
Her i Norge har vi nesten ikke sol, og får derfor ikke tilført de nødvendige 20.000 iu /dag, slik sola gir oss. Vi trenger derfor høyere verdier på vit-D enn det som er max lov å kjøpe i Norge, ellers blir vi lett syke.
22 April 2016 at 7:16 am
Stop taking our freedom away!!!!!!!
22 April 2016 at 7:22 am
Listen to the evidence and do not limit vitamin D intake. It is a supplement proven to be highly cost effective and reduces risk in a wide range of conditions.
http://www.vitamindcouncil.org/about-vitamin-d/testing-for-vitamin-d/
22 April 2016 at 7:33 am
Vitamin D is a proven supplement that is highly cost effective and reduces the risk of a wide range of conditions including various cancers, heart attack, hypertension, diabetes, osteoporosis and bone fractures.
The target levels for circulating 25-hydroxyvitamin D for adults should be set at 125 mmol/L (50 ng/ml), in line with the recommendations of the Vitamin D Council and an extensive Polish study.
EFSA should follow the reference ranges for circulating 25-hydroxyvitamin D given by the Vitamin D Council, which are as follows:
Deficient: 0-100 nmol/l (0-40 ng/ml)
Sufficient: 100-200 nmol/l (40-80 ng/ml)
High Normal: 200-250 nmol/l (80-100 ng/ml)
Undesirable: > 250 nmol/l (> 100 ng/ml) Toxic: > 375 nmol/l (> 150 ng/ml)
24 April 2016 at 5:32 pm
Please do not accept these lowfigures. I taket 5000 per day.
25 April 2016 at 1:25 pm
I am taking 60 000IU per day accompanied with more than 2.5l water/day (to avoid hypercalcemia), closely followed by an MD. That dose is my therapy for MS. Vit D3 is the only efficient treatment for all autoimmune diseases. At the moment there are 5000 CURED MSers living their lives with no impediments or fear of becoming disabled thanks to vit D3, and that number is growing as we speak. To impose any limits on it would be a crime against humanity. Here is the link about the vitamin D3 protocol I'm talking about, it's designed by dr. Cicero Galli Coimbra, a Brazilian neurologist:
https://www.youtube.com/watch?v=IDgoUZAaYmI
25 April 2016 at 6:22 pm
This is yet another means or reducing population in my opinion! Not only are governments geoengineering our climate so that we rarely have clear skies and sunshine - now they plan to reduce our levels of Vitamin D by limiting supplements - already we have rickets back in the UK with Rhondda Cynon Taff in Wales the highest in the country! Mental health problems are through the roof and immunity is at an all-time low with cancer now 1 in 2! All of these problems could be avoided if we were allowed our natural right to sunshine or at least to obtain supplements when we are denied vital Vitamin D via sunshine!
27 April 2016 at 11:23 am
I take 5000ui a day and I double it over the winter, my children also take 2000ui a day and we are never ill, no colds nothing, bags of energy. My partner takes no medication for his arthritis since he has been taking vitamin D 5000iu per day and he also no longer has the frequent infections that he used to have. I am a natural health practitioner and all my clients are taking therapeutic doses of vitamin D and I have seen some incredible health transformations. We need vitamin D, we cant get all we need from the sun we need to supplement it is essential for the body, the immune system, the endocrine system all need it. Suggest that EFSA should follow the reference ranges for circulating 25-hydroxyvitamin D given by the Vitamin D Council, which are as follows:
Deficient: 0-100 nmol/l (0-40 ng/ml)
Sufficient: 100-200 nmol/l (40-80 ng/ml)
High Normal: 200-250 nmol/l (80-100 ng/ml)
Undesirable: > 250 nmol/l (> 100 ng/ml) Toxic: > 375 nmol/l (> 150 ng/ml)
anything else is just too low.
freedom of choice.
Your voice counts
We welcome your comments and are very interested in your point of view, but we ask that you keep them relevant to the article, that they be civil and without commercial links. All comments are moderated prior to being published. We reserve the right to edit or not publish comments that we consider abusive or offensive.
There is extra content here from a third party provider. You will be unable to see this content unless you agree to allow Content Cookies. Cookie Preferences